Scrubs Magazine for Nurses: The Nursing Blog for Healthcare Professionals
Welcome to Scrubs Magazine, the best place to find tips and resources for nurses. Whether you’re a doctor, student, veterinarian, dentist, or on your way to earning your license, we are here to support and educate healthcare providers just like you. From the latest nurse news and information to wild nursing stories that are just too crazy to believe, you’ll find it all right here on Scrubs Mag.
Nurses working today are busier than ever. Between long hours at the hospital and taking care of your loved ones, you probably don’t have time to read through hours of news and stories. Get all the information you need to know right here at Scrubs Mag. Whether you’re looking for the latest heath news, information about travel nursing, or something silly to lighten your day, we’ve got you covered.
Continuing education is just another part of being a provider. You’re never too experienced as a nurse to learn something new. The field of medicine continues to evolve and so should your nursing career. From new regulations and changing health policy to innovative treatments, we will never be the same after the COVID-19 pandemic, but we’re here to help you make sense of everything that’s coming your way.
At Scrubs Mag, we’re no strangers to social media. We follow all the latest trends and pass them onto our readers, so your Instagram feed isn’t filled with noise. Whether you’re looking for fashion tips or a new pair of nursing scrubs, our blog is here to keep you in the loop every step of the way. Find us on Facebook, Twitter, and Instagram to see what’s going in your field.
Never miss a beat with Scrubs Magazine, your trusted source for nursing news.















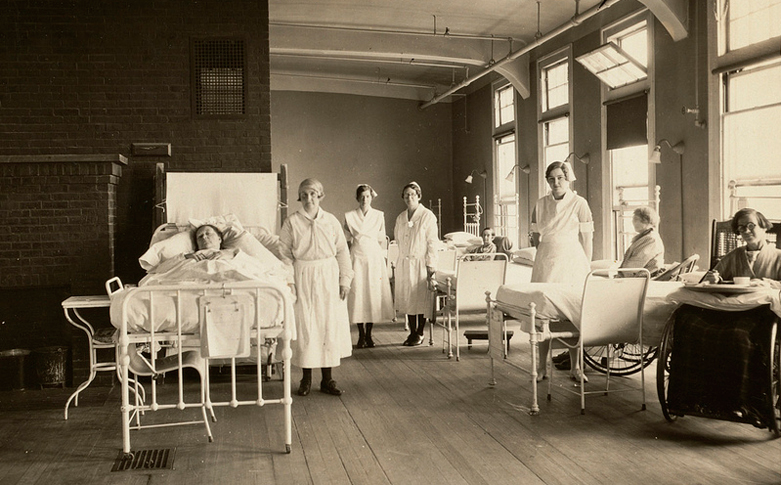 A List Of Rules For Nurses… From 1887
A List Of Rules For Nurses… From 1887
 The Lululemon Of Nursing Scrubs? Everything You Need To Know About The Infinity And Luxe Sport Collections From Cherokee Uniforms!
The Lululemon Of Nursing Scrubs? Everything You Need To Know About The Infinity And Luxe Sport Collections From Cherokee Uniforms!